- Hras
We study the structures of Hras-GTP and Hras-GDP complexes in water solvents in order to investigate the mechanism of the hydrolysis of GTP in Hras-GTP. Understanding the hydrolysis of GTP in Hras-GTP play a key role on overcoming the human tumor. We performed the molecular dynamics simulations of Hras-GTP and Hras-GDP complexes. We evaluated the first hydration spheres of water molecules around PG in GTP and PB in GDP from the trajectories of MD simulations. We also calculated the angular distribution of water molecules around PG in GTP and PB in GDP in the first hydration spheres. The total number of water molecules in the first hydration sphere around PB is larger than the total number around PG. The density of water molecules around PG is higher than the density around PB, in the area: 0° < θ < 30°, -60° < φ < 60°. This difference suggests that GTP hydrolysis in Hras-GTP is triggered by a water molecule in this area attacking γ-phosphate.
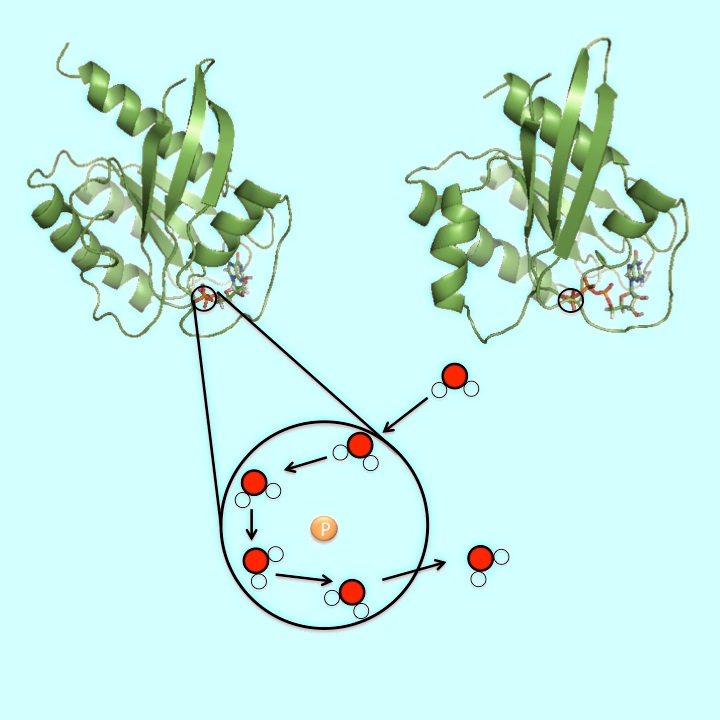
We estimated the first hydration sphere of water molecules around PG in Hras-GTP and around PB in Hras-GDP. We calculated the duration time of a water molecule in the first hydration sphere.
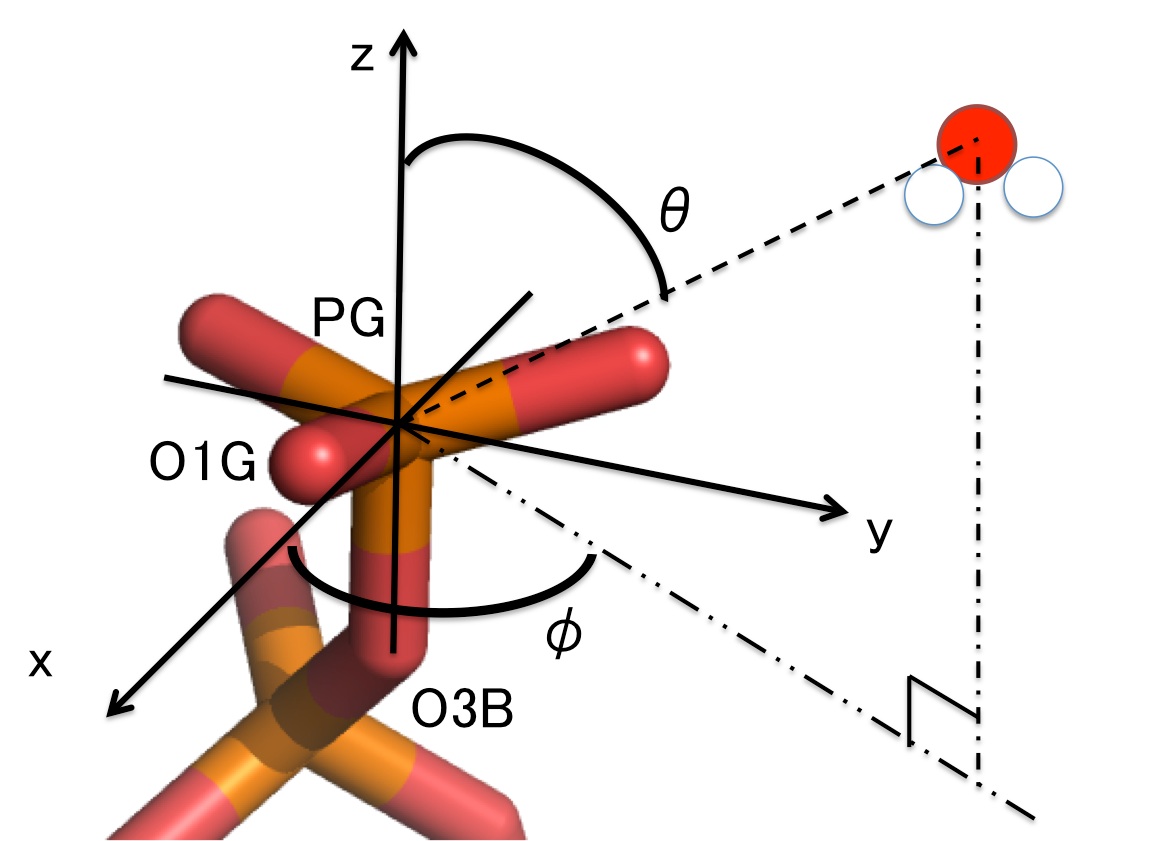
We calculated the angular distribution of water molecules around PG in Hras-GTP and around PB in Hras-GDP using the coordinates defined along the guanine nucleotide.
- Protein Interaction
Design of protein surface to create the functional structure is still a significant challenge in protein engineering. The objective of our study is to design the binding interfaces on proteins surfaces that form protein-protein interaction. We designed several binding interfaces involving some charged and hydrophobic amino acid residues. Molecular dynamics (MD) simulations were performed to explore suitable binding interfaces. We simulated the binding of the protein I (green) and the protein II (blue) (Fig.1 (a)). MD simulation showed that the protein-protein interaction is formed (Fig.1 (b)) depending on the number of charged and hydrophobic amino acid residues in the binding surfaces.
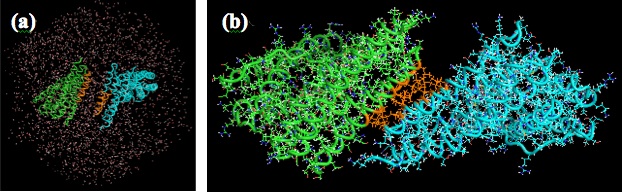
Fig.1 (a) The initial state of the MD simulation. (b) The binding state of two proteins.
- Laminin
The peptides derived from laminin, a vital glycoprotein, have various biological activities, and can be applied to pharmaceutical agents and biomaterials. We perform conformation analysis of peptides derived from laminin by computer simulation (molecular dynamics simulation). Thus, conformation analysis of these peptides is a study of significance, and our results may help the study of pharmaceutical agents and biomaterials.
- Glycoproteins
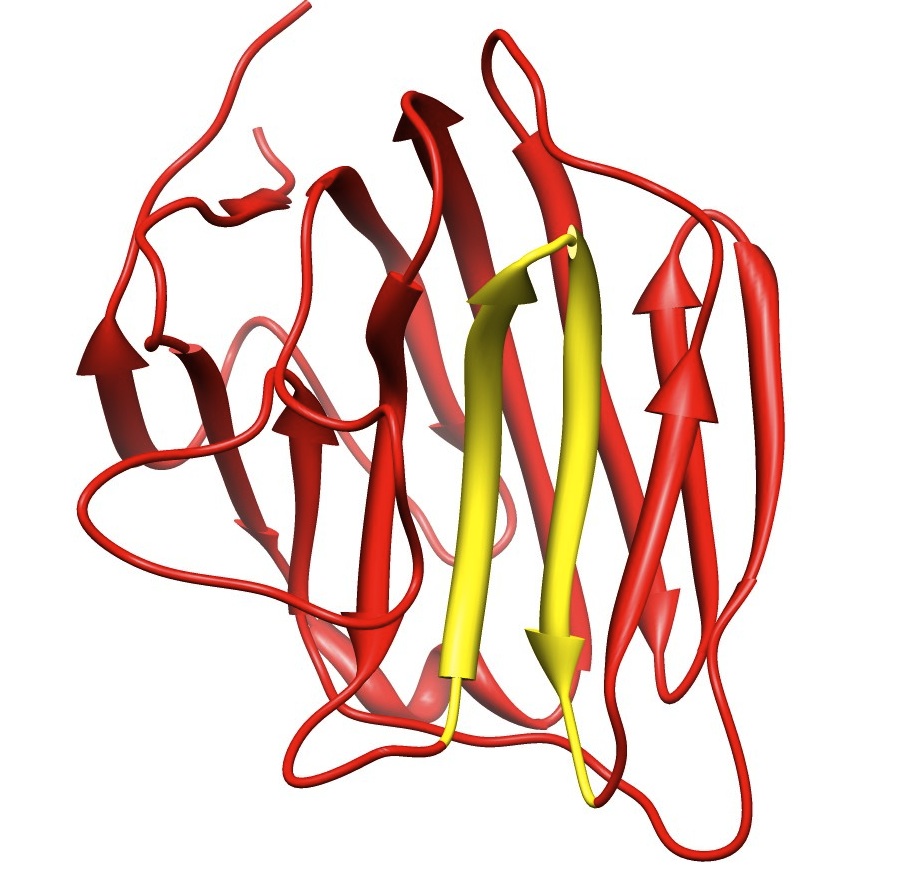
We investigate glycoproteins which are part of basement membrane. Glycoproteins have diverse biological functions, such as cell adhesion, migration and angiogenesis. They are also essential for proper muscle function. Lack of glycoproteins may lead to muscular dystrophies, characterized by progressive skeletal muscle weakness, defects in muscle proteins, and the death of muscle cells and tissue. We perform molecular dynamics simulation of glycoproteins to compare with crystal structure obtained by X-rays.
- The Incretins
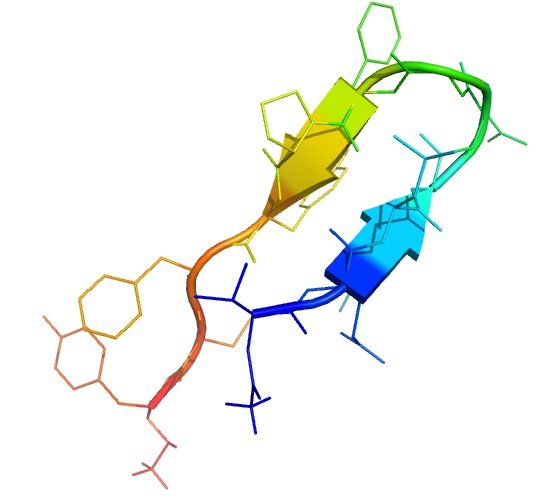
The incretins are gut hormones which function as stimulators of insulin secretion from the pancreas. We investigated the structure of the incretins associated peptide used for the treatment of diabetes by molecular dynamics simulation. We study the effect of solvents, and analyze the influence on the structure. Furthermore, We are interested in reproducing the peptide dynamics in the body by changing the osmotic pressure.


MD simulation at 300 K in water (left) and in 35% TFE (right).
- Bacterial Colonies
We theoretically study patterns and statistical properties of bacterial colonies by means of computer simulation. From the experimental study, Bacillus subtilis, which switches the direction of rotation of their flagella and change their motile mode between running and tumbling, is known to exhibit various colony patterns (DLA-like, Eden, DBM-like, homogeneous disk-like and concentric ring-like) depending on the substrate softness and nutrient concentration. Another bacteria species Thermus thermophilus, which is genetically and phenotypically known to have TFP and shows twitching evidence, forms a colony with wriggling contour of edge. We notice the various motility characteristics of the bacteria and propose several microscopic models for a bacterial individual. These models are analyzing from the viewpoint of physics and mathematical science and the growth properties of bacterial colony is investigated by computer simulation. We have shown that not only the difference in the environment of a colony but the difference in the motility characteristic has determined the form of a colony.
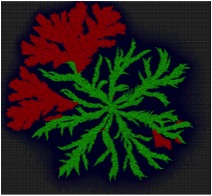
(Fig.1) The colony consists of two species of bacteria individuals.

(Fig.2) The colony formed by the twitching motility shows wriggling contour of edge.
- Polymer Gel
Polymer gel has three-dimensional network structure consisting of polymers. Hydrogel are used in various products such as medical supplies, and many possibilities of the applications are expected in the future. Since it is difficult to observe the gel formation process and structure of the tetra-polyethylene glycol (tetra-PEG) gel in the experiments, we perform molecular simulation.
We construct a coarse-grained model of the tetra-PEG for our molecular dynamics simulation. Extending Hashimoto’s model consisting of five parts, we use a model of nine parts. We use Lennard-Jones potential, spring potential and angle potential. Based on the results of such simulation and comparison with the results of study by Hashimoto, we discuss the gel formation process and structure of the tetra-PEG gel.

The state before the reaction for the case of three hundred monomers is shown.